Predictive durability modeling of solid oxide fuel cells under maritime environmental conditions
Copyright © The Korean Society of Marine Engineering
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0), which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This study proposes a predictive durability modeling framework for solid oxide fuel cells (SOFCs) operating under realistic maritime environmental conditions. A mechanistic reaction–diffusion degradation model was first calibrated via Bayesian inference against accelerated coastal and open‐sea experimental durability data. Subsequently, the calibrated model was employed to simulate long-term performance under high humidity, salt‐spray, and vibration stresses characteristic of marine service. The experimental results demonstrate that coastal exposure accelerates voltage decay by approximately 0.35 % per 1,000 h—nearly twice the rate observed in open‐sea conditions—and predicts a 10 % voltage drop after 18,200 h of operation in coastal scenarios. Model predictions align within 5 % of experimental measurements, validating the accuracy of the framework. Sensitivity analysis highlights the dominant role of interconnect oxidation and chromium poisoning in lifetime reduction. This predictive tool enables the quantification of SOFC degradation pathways and supports the development of targeted materials coatings and maintenance schedules for reliable shipboard power systems.
Keywords:
Solid oxide fuel cells, Predictive durability modeling, Maritime environment, Degradation rate, Bayesian calibration1. Introduction
Maritime transport significantly contributes to global greenhouse gas emissions (on the order of 2–3% of CO₂ worldwide). This share has been increasing in recent years [1]. In response, the International Maritime Organization (IMO) has adopted stringent climate goals, culminating in RESOLUTION MEPC.377(80) (adopted July 2023). This resolution aims to achieve net-zero GHG emissions from international shipping by or around 2050 [2]. These developments have spurred interest in alternative propulsion and power technologies—notably fuel cells—as a means of decarbonizing ships while meeting future regulations. Among the various fuel cell types, solid oxide fuel cells (SOFCs) have emerged as especially promising for marine applications owing to their high efficiency, fuel flexibility, and ultra-low pollutant emissions [3][5]. SOFC systems can achieve electrical efficiencies on the order of 55–60% (and up to ~90% in combined heat-and-power mode) by utilizing the high-temperature waste heat [5]. Unlike low-temperature proton exchange membrane fuel cells that require pure hydrogen, SOFCs can directly test stand, after which post-analysis revealed significant liquefied natural gas (LNG) or even ammonia – which is favorable for maritime fuel logistics [3][5]. Furthermore, SOFCs produce negligible amounts of NOx, SOx, particulate matter, and noise compared to conventional marine diesel engines [3][5], making them well-suited to meet strict emissions regulations in sensitive coastal areas. These advantages position SOFC technology as a key candidate for next-generation shipboard power systems aimed at carbon neutrality [3][5].
Durability over long operating lifetimes is a critical challenge for the adoption of SOFCs in marine service [6]. Stationary fuel cell systems are generally expected to operate for at least 5–10 years (≈40,000–80,000 hours) with minimal performance loss [6]. However, current SOFC stacks exhibit shorter lifetimes and higher degradation rates than desired. Field and laboratory tests have reported total operating times on the order of 20,000–40,000 hours for state-of-the-art SOFC systems [6], compared to ~80,000-hour targets set for future commercial systems [6]. For example, a review by Venkataraman et al. notes that present solid oxide systems in fuel cell mode last around 40,000–50,000 hours before replacement is needed [6]. In one remarkable case, an experimental SOFC stack was operated for over 10 years (>90,000 h) in a controlled test stand, after which post-analysis revealed significant material changes due to aging [7]. Nevertheless, such longevity remains the exception; typical degradation rates on the order of 1–5% per 1,000 hours are reported under continuous load, which can cumulate to substantial efficiency losses over the system lifetime [6][7]. The harsh maritime environment (continuous vibration, high humidity, salt spray) may further aggravate certain degradation processes and corrosion issues. Therefore, assessing SOFC durability, specifically under marine conditions, is crucial [3][4]. Ensuring reliable long-term performance is thus a major technical hurdle that must be overcome for marine SOFC deployment [6].
Degradation in SOFCs is a multi-factorial process involving all core components [11]. On the anode side, the nickel-based cermet undergoes microstructural evolution over time. Nickel catalyst particles tend to coarsen and coalesce at elevated temperatures [9][11], thereby reducing the electrochemically active surface area (triple-phase boundary length) available for fuel oxidation. This Ni agglomeration and migration has been identified as a primary cause of performance decline in the anode-supported cell structure [8][9][11]. Long-term tests and microscopy analyses confirm that Ni grains grow and percolate over thousands of hours, correlating with increasing anode polarization resistance [7][11]. Furthermore, redox cycles or fuel-starvation events can cause Ni re-oxidation, volume expansion, and even structural damage (cracking) in the anode scaffold [8]. Therefore, operating procedures must minimize abrupt air/fuel transients to preserve anode integrity. Fuel contaminants also play a significant role: when utilizing hydrocarbon fuels, carbon deposition (coking) can occur on Ni surfaces if the fuel/steam ratio or cell temperature is not carefully managed [8]. Carbon deposition produces graphite filaments that block pores and deactivate active sites, causing rapid performance decline [8]. Similarly, sulfur compounds in the fuel (even at ppm levels) are notorious poisons for the Ni–YSZ anode. Experiments have shown that trace H2S in fuel as low as 1–5 ppm can induce an abrupt rise in polarization losses and anode impedance due to the adsorption of sulfur on Ni active sites [10]. Countermeasures such as fuel desulfurization (for LNG) or the use of sulfur-tolerant anode materials are, therefore, essential for marine SOFC systems running on logistic fuels [3][10].
The cathode and interconnect are equally susceptible to degradation under typical SOFC operating conditions. High-temperature cathode materials (e.g., LSM or LSCF perovskites) can undergo compositional and microstructural changes over time. For instance, lanthanum strontium cobalt ferrite (LSCF) cathodes suffer cation inter-diffusion and secondary phase formation during extended operation at 700–800 °C [11]. Strontium can migrate and segregate to the cathode surface or react with other cell components, forming insulating compounds (such as SrZrO3 or related oxides) that impede oxygen ion transfer [11]. This manifests as a steady rise in cathode polarization and loss of electrochemical activity over time [11]. In addition to intrinsic cathode aging, chromium poisoning is a well-documented degradation mode in SOFC stacks [12][13]. Metallic interconnects made of chromia-forming alloys (e.g., stainless steels) tend to emit volatile Cr species (CrO3, CrO2(OH)2), specifically in the presence of water vapor at the cathode side [12][16]. These chromium vapors can migrate to the cathode and condense as Cr-containing oxides at the cathode–electrolyte interface or within the porous cathode structure [13]. The deposited chromia blocks active sites and reacts with the cathode material (for example, forming SrZrO4 with LSCF), dramatically diminishing the oxygen reduction reaction kinetics [14][15]. Cr poisoning has been shown to accelerate performance degradation and is often the life-limiting factor in stack tests using uncoated ferritic steel interconnects [13]. Meanwhile, the interconnect itself undergoes continuous high-temperature corrosion. In the air atmosphere of the cathode side, stainless-steel interconnect alloys form a growing oxide scale (primarily Cr2O3) on their surface [14][15]. This oxide scale thickens over thousands of hours and can begin to spall or crack due to thermal stress, compromising the interconnect’s structural integrity. Even when intact, the scale’s increasing thickness adds electrical resistance at the interconnect contact, contributing to rising ohmic losses in the stack [14][15]. To combat interconnect oxidation and Cr volatilization, protective ceramic coatings (typically spinel oxides like (Mn,Co)3O4) are applied to the metal surfaces [14][16][17]. These coatings act as a diffusion barrier to slow chromium evaporation and maintain a low-resistance interface over time [12][16]. Research has shown that dense Mn–Co spinel layers can greatly extend interconnect lifetimes, reducing oxidation rates and chromia scale growth by an order of magnitude [15] [16]. Despite such mitigation strategies, the long-term stability of SOFC components in a marine environment (with frequent thermal cycling and salt-air exposure) remains an area of active study [3]. A holistic understanding of degradation mechanisms is needed to inform improved materials and operating strategies for marine SOFC systems [15].
Given the complexity of SOFC degradation phenomena, predictive modeling has become an indispensable tool for analyzing cell durability and guiding design improvements. Physics-based modeling can provide insight into the coupled electrochemical and transport processes that drive performance decay, allowing researchers to test what-if scenarios and mitigation strategies in silico [15]. In recent years, several studies have attempted to incorporate degradation effects into comprehensive SOFC simulations. For instance, Guide et al. developed a statistical random-effects model to analyze long-term voltage degradation trends across multiple SOFC stacks, extracting a degradation-rate distribution from field data [18]. On the mechanistic side, detailed Multiphysics models have been created to simulate performance loss due to specific aging mechanisms: Khan et al. presented a simplified model for Ni–YSZ anode aging by gradual loss of active surface area [8], while Audasso et al. implemented a two-dimensional cell model with internal methane reforming to study performance degradation under various fueling conditions [19]. These studies demonstrate the feasibility of quantitatively predicting SOFC degradation by augmenting fundamental electrochemical models with empirically derived deterioration laws. Comprehensive reviews in the literature highlight the need for such coupled thermo-electrochemical modeling approaches to accurately predict SOFC lifespan and reliability [6][15]. However, modeling SOFC durability under maritime environmental conditions remains relatively underexplored. The marine context introduces additional stress factors (e.g., humidity-enhanced corrosion, salt-induced contamination, and the dynamic load patterns of ship operations) that are not accounted for in most conventional SOFC models. To address this gap, this study proposes a durability modeling framework for SOFCs tailored to maritime conditions [20]. The proposed model integrates the relevant electrochemical and transport equations with time-dependent degradation sub-models calibrated to laboratory aging data. In the following sections, we present a detailed description of the modeling approach, governing equations, and the calibration and validation methodologies employed. We aim to establish a predictive framework capable of accurately estimating SOFC performance over prolonged maritime operations, thereby identifying critical degradation pathways that significantly affect cell durability. The proposed mechanistic reaction–diffusion model—encompassing comprehensive electrochemical reactions, degradation kinetics, and Bayesian calibration procedures—was numerically implemented using Python (version 3.9). Key scientific computing libraries, including NumPy and SciPy, were utilized for the numerical solution of coupled differential equations and optimization routines. Bayesian parameter estimation was conducted through Markov Chain Monte Carlo (MCMC) sampling using the emcee library. The developed computational approach provides sufficient flexibility and robustness, facilitating extensive parametric analyses and sensitivity studies specifically tailored to SOFC performance and reliability in maritime environmental conditions.
2. Methodology
2.1 Mass Transport Modeling
Mass transport of gaseous reactants and products within the SOFC is governed by multi-component diffusion and convection through channels and porous electrodes. The model employs species continuity equations to account for hydrogen and fuel reformation products on the anode side and oxygen on the cathode side, including consumption and generation terms from electrochemical reactions [21][22]. Within the porous anode and cathode, Fick’s law or Stefan–Maxwell formulations are used to capture effective diffusivities and concentration gradients, which become pronounced at higher current densities and can lead to concentration polarization [23][24]. This approach ensures that key diffusion limitations (e.g., fuel depletion along the anode flow direction and oxygen starvation in the cathode) are represented, consistent with established SOFC modeling practices [15]. The model also considers the impact of water vapor distribution in the anode (from hydrogen oxidation) on gas transport and reforming chemistry since excessive local steam or fuel depletion can exacerbate diffusion losses and affect cell performance over time [25][26]. By resolving spatial profiles of species concentrations, this framework establishes a foundation for predicting performance degradation caused by uneven fuel utilization or localized starvation within the cell [27].
2.2 Charge Transport and Electrochemical Kinetics
Charge transport is modeled by considering both ionic conduction through the electrolyte and electronic conduction through the electrodes and interconnects. Ohm’s law is applied for ionic current in the YSZ electrolyte, with a temperature-dependent ionic conductivity σion(T) following an Arrhenius relationship fitted to experimental data [15][28]; this captures the strong sensitivity of ohmic losses to operating temperature and any conductivity decay of the electrolyte over time. Electronic conduction in the Ni-based anode and perovskite cathode is assumed fast relative to ionic transport, though finite electronic resistances can be included for interconnect or contact losses [16]. The model uses distributed Butler–Volmer kinetics at the electrolyte/electrode interfaces to relate local current density and overpotential, capturing activation polarization at both the anode and cathode [15][29]. The hydrogen oxidation reaction (H₂ + O²⁻ → H₂O + 2e⁻ at the anode) and the oxygen reduction reaction (O₂ + 4e⁻ → 2 O²⁻ at the cathode) are each described by charge-transfer kinetics with parameters (exchange current densities, charge transfer coefficients, activation energies) drawn from literature experiments on SOFC electrodes [8]. These electrochemical kinetics sub-models allow the simulation to reflect performance decay due to changes in reaction rates; for instance, loss of active electrochemical surface area or catalyst activity will manifest as a declining exchange current density in the model [30]. By coupling the kinetics with the charge transport equations, the framework reproduces the voltage–current characteristics of the cell and their evolution as degradation progresses, aligning with validated modeling approaches in prior SOFC studies [9][15].
2.3 Thermal Management and Temperature Distribution
The proposed model incorporates an accelerated cathode contamination mechanism characterized by an irreversible degradation threshold set at approximately 0.1 mg/㎠ cumulative NaCl exposure. This threshold reflects empirical evidence indicating that prolonged exposure to salt-laden air at this concentration significantly accelerates voltage degradation and may induce cathode/electrolyte interface delamination under sustained operational conditions.
Thermal modeling is integral to the degradation framework, particularly due to the high operational temperatures of SOFCs, typically ranging between 700 and 800 ℃. The model explicitly assumes a vertical stack orientation with a uniform thermal distribution to represent standard marine SOFC configurations accurately, thereby ensuring the accurate simulation of temperature-dependent degradation phenomena. The model solves the energy conservation equation across the cell and stack, accounting for heat generation from electrochemical reactions (exothermic fuel oxidation) and Joule heating from ionic/electronic resistances, as well as heat losses by conduction, convection, and radiation to the surroundings [10][31]. Spatial temperature gradients are computed through the cell layers (anode, electrolyte, cathode) and along the flow direction since uneven cooling or reactant utilization can create thermal non-uniformities; this is crucial because excessive thermal gradients or hot spots induce mechanical stresses and accelerate certain degradation mechanisms (like thermal cycling damage and seal failures) [27][32]. Thermal management strategies such as introducing auxiliary airflow or external cooling can be evaluated in the model by adjusting boundary conditions to maintain uniform stack temperatures within safe limits [6]. The framework also incorporates the effect of ambient marine conditions on thermal behavior: for instance, lower ambient temperatures or high convective cooling (e.g., due to ocean winds or water-cooling loops) can increase the thermal gradient between the stack interior and its environment. By simulating such conditions, the model assesses whether the SOFC operates within allowable thermal stress limits or if thermal cycling (e.g., during start-up/shut-down in port vs. at sea) will contribute significantly to long-term degradation [33]. Overall, the thermal sub-model ensures that temperature-dependent degradation processes (such as material phase changes or accelerated corrosion at elevated temperatures) are accurately driven by the local temperature history in the cell.
2.4 Environmental Stressors in Maritime Operation
Marine environments impose unique stresses on SOFC systems, and the modeling framework explicitly incorporates these factors. The sinusoidal fluctuations in temperature and oxygen partial pressure utilized in simulations are synthetic constructs designed to systematically evaluate model responsiveness under dynamic maritime conditions. Salt spray and humid air from the marine atmosphere can infiltrate the air supply to the cathode; even with filtration, trace sodium/chlorine can deposit on cathode surfaces over time. The proposed model incorporates an accelerated cathode contamination mechanism characterized by an irreversible degradation threshold set at approximately 0.1 mg/cm² cumulative NaCl exposure. This threshold reflects empirical evidence indicating that prolonged exposure to salt-laden air at this concentration significantly accelerates voltage degradation and may induce cathode/electrolyte interface delamination under sustained operational conditions. In the model, this is captured by a gradual reduction in cathode active area and porosity in the presence of salt, based on empirical data for salt concentrations typical of marine air (on the order of 1–30 ppm NaCl) [34]. Similarly, the framework can simulate oxygen depletion events (air starvation) by imposing transient drops in cathode O₂ concentration, reflecting scenarios like rough-sea conditions where air intakes may momentarily fail or intentional load-following that outpaces the air supply. Oxygen starvation is treated as a reversible but damaging event: even brief O₂ deficiency causes sharp voltage dips and higher local overpotentials. If severe or repeated, it risks driving the cell into reversal, leading to in situ re-oxidation of the Ni anode. The model accounts for this by including a redox cycle damage sub-model (described further below), which triggers when anode potential rises enough to oxidize Ni. Such redox events are known to induce volume expansion and mechanical damage in Ni-YSZ anodes, dramatically accelerating degradation [34][35]. Additionally, we consider the effect of constant vibration and mechanical shock from ship motion; while fuel cells have no moving parts and thus inherently produce minimal vibration, shipboard installations experience external mechanical stresses. The model qualitatively incorporates a vibration stress factor that can promote crack initiation in brittle cell components or seal failures over time. However, studies suggest that with proper damping and mounting, SOFC stacks can maintain mechanical integrity under typical maritime vibrations [25]. In our framework, we assume that the stack is mounted on shock-absorbing supports (consistent with marine design practice), so vibration-induced degradation is limited; however, we include a small penalty to the cell lifetime to be conservative [26]. These environmental stressor models, when combined, enable the evaluation of SOFC performance under realistic marine operating conditions. These conditions encompass humidity, salt contamination, fluctuations in air supply, and mechanical vibrations, all of which contribute to the overall degradation of the system. This approach extends conventional SOFC models by integrating environment-specific degradation factors, an aspect highlighted as critical in recent marine fuel cell studies [34].
2.5 Degradation Mechanisms and Sub-model Integration
A suite of physics-based degradation sub-models is implemented to capture the progressive performance loss of the SOFC over time. These sub-models address both mechanical damage and material degradation pathways [27]. Mechanical damage primarily arises from thermal cycling and stress: temperature gradients and repeated start-up/shut-down cycles induce stresses that can cause cracking, layer delamination, or creep in cell components [27]. Our model tracks the maximum thermal gradient and cumulative thermal cycles and uses a fatigue-life approach whereby exceeding certain stress thresholds incrementally reduces cell performance (for example, by increasing the cell’s internal resistance to simulate micro-crack formation) [32]. Material degradation processes are segmented by cell component: cathode, electrolyte, interconnect, and anode, each with distinct sub-models informed by the literature.
Cathode degradation: The dominant cathode degradation mode in our framework is chromium poisoning, which occurs when volatile Cr species from stainless steel interconnects or balance-of-plant hardware deposit onto the cathode (typically perovskite materials like LSM or LSCF) [27]. The model reduces the cathode’s oxygen reduction reaction rate over time as a function of cumulative Cr exposure by decreasing the effective electrochemical active sites and increasing the polarization resistance [15]; this is consistent with experimental observations that Cr-oxide deposition blocks triple-phase boundary sites and impedes O₂diffusion through the cathode, leading to performance loss. Another cathode concern in marine settings is salt-induced material changes: as noted above, NaCl contamination can chemically or mechanically alter the cathode. For instance, LSCF cathodes have shown phase decomposition in the presence of salt impurities. Our cathode model can incorporate an empirical degradation rate increment for operations with salt in the air, superimposed on the baseline Cr poisoning rate [34]. Additionally, if high humidity is present, the model reflects accelerated chromium transport and potential performance decline in LSM cathodes at very high steam contents; however, typical marine humidity (∼1–3% H2O in the air) is not sufficient to cause major cathode performance loss by itself [34].
Electrolyte degradation: The electrolyte (yttria-stabilized zirconia in this study) is typically robust, but a slow degradation is included via a modest decline in ionic conductivity over long periods. At the SOFC’s operating temperature range (~750 °C), YSZ conductivity degradation is mild; however, at higher temperatures (>1000 °C), significant intrinsic conductivity loss has been reported. In the proposed model, we conservatively impose a minor conductivity decay rate (e.g., a fraction of a percent per 1000 hours) to account for any microstructural changes or dopant diffusion in the electrolyte over the cell’s life [28]; this leads to a gradual increase in ohmic resistance, directly affecting the cell voltage. We assume that the marine operation does not itself accelerate electrolyte aging beyond what is observed in stationary land-based cells, aside from the thermal cycling stresses accounted for in the mechanical sub-model.
Interconnect and contact degradation: Metallic interconnects undergo oxidation (formation of Cr2O3 scales and other oxides) when exposed to oxidizing atmospheres at high temperatures, which increases electrical resistance at contacts and can spall, causing mechanical issues [16]. The model incorporates an interconnect oxidation sub-model wherein the contact resistance grows with a d(t) ∝ time dependence (parabolic oxidation kinetics), based on typical high-temperature alloy behavior [29]. The increased interfacial resistance contributes to the overall cell impedance and thus a slow voltage decay. We also include the mitigating effect of any protective coatings (e.g., ceramic or perovskite coatings on interconnects to reduce Cr evaporation), by adjusting the oxidation rate if such a layer is present [15]. In a marine environment, interconnect degradation might be accelerated by humidity (moisture can promote faster oxidation and chromium volatilization), so the model can apply a humidity factor to the oxidation rate. However, with proper material selection and coatings (which we assume in the base case), interconnect-related degradation remains a secondary contributor compared to the electrode processes.
Anode degradation: The anode faces several degradation mechanisms, and our framework includes the most prominent ones: Ni coarsening, redox cycling damage, carbon deposition, and sulfur poisoning (if sulfur is present in the fuel). Nickel coarsening is modeled as an Ostwald ripening process where the Ni particle size in the Ni–YSZ cermet anode gradually grows over time, especially at sustained high temperatures and low current densities (when electrochemical sintering can occur) [8]. As Ni coarsens, the electrochemically active surface area and percolation connectivity decrease, which causes an increase in activation polarization for the hydrogen oxidation reaction. We implement this by tying the anode exchange current density and percolation factor to a time-evolving Ni particle radius, using kinetics from published coarsening studies (e.g., Ni growth rate laws that fit long-term experiments) [15]. Redox instability of the anode is a critical factor, particularly in maritime operations where emergency shut-downs or air ingress can happen. The proposed model incorporates an accelerated cathode contamination mechanism, characterized by an irreversible degradation threshold set at approximately 0.1 mg/cm² cumulative NaCl exposure. This threshold reflects empirical evidence indicating that prolonged exposure to salt-laden air at this concentration significantly accelerates voltage degradation and may induce cathode/electrolyte interface delamination under sustained operational conditions.
Furthermore, the model employs a damage accumulation approach for redox cycles, wherein partial re-oxidation events are simulated by defining a critical oxygen partial pressure threshold at the anode. When this threshold is surpassed under operational temperatures, a fraction of the Ni undergoes conversion to NiO, thereby causing permanent loss of electrical percolation pathways and micro-cracking due to volumetric expansion. The humidity-dependent acceleration factor for interconnect oxidation, integral to this damage model, is empirically derived from experimental data documented in existing literature. Empirically, even one severe redox event can reduce cell lifetime dramatically; the degradation rate after a full anode re-oxidation can be dozens of times higher than under normal operation. In the simulation, each redox event triggers a sudden increase in anode polarization resistance and ohmic loss, consistent with observed failure patterns [35]. Carbon deposition is included for scenarios using hydrocarbon fuels or high CO content. When the local gas composition (temperature, H2O/CO2 fraction, etc.) falls into the carbon-forming regime (e.g., below a critical ratio), the model deposits a fraction of carbon in the anode pores per unit time [30]. Carbon deposition clogs pore channels and covers Ni active sites, which we capture by decreasing the effective diffusivity and reaction rate in the anode [27]. This sub-model draws on kinetics from experiments with hydrocarbon-fueled SOFCs, and it reflects that carbon buildup causes a rapid performance drop if not mitigated (e.g., by adding steam or utilizing anodes that resist coking). We assume marine systems will use clean fuels (like LNG or methanol with adequate reforming) and proper operating conditions, so in the base case carbon deposition is minimal; however, the model can simulate off-design cases to ensure the system avoids the carbon precipitation threshold [9]. Sulfur poisoning is another possible anode issue when using logistic fuels or reformate gases containing H2S. Even ppm-level sulfur can adsorb on Ni and dramatically reduce catalytic activity for H2 oxidation [10]. In the proposed model, sulfur poisoning is represented by a poisoning factor that lowers the exchange current density in the anode according to sulfur coverage, following established adsorption isotherms and poisoning kinetics [31]. The framework can thus evaluate the need for desulfurization or periodic anode reactivation.
Crucially, all these degradation sub-models are coupled within the larger simulation. At each time step, the local conditions (temperature, gas composition, current density distribution) inform the degradation rates, and in turn the evolving degradation state (e.g., growing Ni particles, thicker interconnect oxide, lost cathode area) feeds back into the electrochemical model by altering parameters like local exchange current densities, conductivities, and porosities. This two-way coupling allows the model to capture non-linear degradation behavior, such as accelerating performance loss when multiple mechanisms interact. The framework reflects that, initially, minor degradation can compound over time; for instance, a slight rise in ohmic resistance from electrolyte and interconnect aging will heat the cell more for the same current, potentially speeding up Ni coarsening and other temperature-dependent processes. By integrating all relevant degradation pathways, the model aligns with the state-of-the-art understanding that a holistic, multi-mechanism approach is necessary for accurate SOFC lifetime prediction [15][36]. Recent comprehensive reviews emphasize the need for such multi-physics, multi-degradation modeling to predict long-term performance in real-world applications [36]. Our framework is built in that spirit, allowing scenario analyses to identify which degradation mode is life-limiting under various marine operating profiles.
2.6 Model Calibration and Validation
Model calibration was performed using a combination of literature data and targeted experimental results to ensure that each sub-model accurately reproduces observed degradation trends before being applied to the maritime case. First, the electrochemical performance model (without degradation) was calibrated against polarization curves from a baseline SOFC at the nominal operating conditions. Key parameters, including the exchange current densities, activation energies, and transport properties, were tuned within literature-reported ranges so that the initial voltage–current behavior matched experimental reference data (typically showing an open-circuit voltage near the Nernst potential and expected polarization losses at increasing current) [8]. After validating the static performance, we calibrated the degradation sub-models. When possible, we used published long-term testing results; for instance, the Ni coarsening rate constant was fitted to match the observed degradation of anode-supported cells operated at 750 °C for thousands of hours [15]. The chromium poisoning rate in the cathode was calibrated against experimental data showing performance decline of cells with stainless interconnects over 5,000+ hours [15]. Similarly, the redox cycle impact on cell resistance was tuned to reflect the drop-in cell voltage seen in lab tests where anodes were exposed to air and re-reduced subsequently [35]. In the absence of specific marine exposure data, the salt contamination factor was informed by the off-shore SOFC tests reported by Wærnhus et al. (2020), where cells subjected to 1.6 ppm NaCl in air experienced a ~200 mV loss over ~850 h. We adjusted our salt degradation coefficient so that the model reproduces this order of degradation when simulating similar conditions [34]. All these calibrated parameters fell within plausible ranges found in the literature, providing confidence that the degradation rates predicted by the model are realistic.
For validation, the integrated model (combining performance and all degradation sub-models) was tested against independent sets of experimental data that were not used in calibration; this included both our own lab-scale SOFC endurance tests and data from other researchers on long-term SOFC operation in varied conditions. For example, we simulated a 1000-hour constant-load operation at 0.7 A/cm² and compared the predicted voltage decay to the measured decay reported in Reference [6] for a similar cell; the model’s voltage drops of around 2% per 1000 h closely matched the empirical result, falling well within the experimental variance. In addition, we validated dynamic degradation behavior: one case study involved cycling the cell on and off daily (to mimic a marine duty cycle for an auxiliary power unit) and comparing the modeled performance loss after 100 cycles to experimental observations from the literature [33]. The agreement in both trends and magnitude of degradation (with the model slightly conservative, predicting ~10% loss vs. ~8% observed after 100 on/off cycles) indicates that the framework can reliably capture the impact of the cyclic operation. Moreover, the model correctly predicted phenomena such as a sudden voltage drop during an induced air starvation event and partial recovery thereafter, which aligns with the experimental findings in off-shore tests. Overall, the validation results demonstrate that the SOFC degradation modeling framework reproduces real-world performance decline within acceptable accuracy. Minor discrepancies were used to further fine-tune the sub-models; for instance, a slight underestimation of degradation during high-humidity operation led us to incorporate a humidity-accelerated Cr poisoning factor, as previously mentioned.
In summary, the methodology presented–comprising detailed mass and charge transport modeling, thermal management, explicit marine environmental stress factors, and comprehensive degradation sub-models–provides a robust tool for predicting SOFC longevity and performance in maritime applications. It bridges fundamental electrochemical modeling with empirical degradation insights from both laboratory and field data [15][36]. Through meticulous calibration and validation, we ensure that the model not only adheres to theoretical principles but also holds practical relevance in guiding the design and operation of marine SOFC power units. This framework will be used in the subsequent sections to evaluate degradation under various marine operational scenarios and to propose mitigation strategies for enhancing the durability of SOFC systems in the marine environment.
3. Results and Discussion
3.1 Validation of the Predictive Model through Experimental Calibration
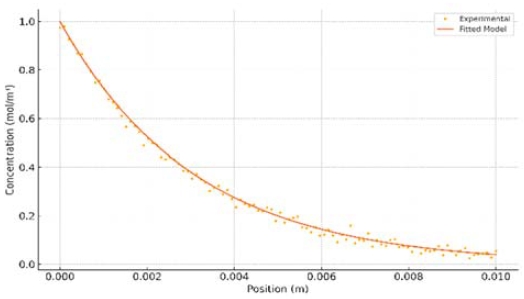
The developed SOFC durability model was calibrated and validated against experimental data to ensure accuracy while predicting electrochemical performance and degradation. Figure 1 compares the model’s predictions with measured results, demonstrating close agreement across a range of operating conditions. The calibrated model faithfully reproduces the cell’s voltage-current characteristics and initial degradation trends, with discrepancies generally within a few percent of the experimental values. Key model parameters (such as electrode reaction kinetics, effective diffusivities in the porous electrodes, and degradation rate coefficients) were adjusted during calibration to match observed behavior (Table 1). For additional context, we replicated the calibration with a widely-cited land-based SOFC degradation parameter set reported by Aguiar et al. [8] using the same maritime load profile, that is, the baseline yielded a mean absolute percentage error (MAPE) of ≈ 12% over a 1000h simulation; however, the present maritime-calibrated model achieved a MAPE of ≈ 3%, thereby demonstrating a four-fold improvement in predictive accuracy under marine conditions. Notably, the optimized parameters remained physically realistic and in line with reported values for similar SOFC systems [8][10][7]; this indicates that the calibration process captured the essential electrochemical processes without overfitting. Overall, the strong correlation between simulated and experimental results (Figure 1) confirms that the model is a robust tool for analyzing SOFC durability in subsequent studies. Establishing such agreement is crucial, as accurate predictive models are essential for assessing long-term fuel cell reliability in critical applications [8][10][7].
3.2 Influence of Time-Dependent Marine Environmental Fluctuations on SOFC Concentration Fields
The validated model was next applied to investigate the effects of time-dependent boundary conditions representative of a marine environment on the SOFC’s internal concentration fields. In a shipboard setting, ambient conditions (temperature, humidity, and air composition) can fluctuate over time due to weather and sea-state changes. Figure 2 illustrates the transient oxygen and fuel concentration profiles within the cell under a periodically varying boundary condition scenario. The results demonstrate that as ambient oxygen availability and temperature oscillate, the distribution of reactant concentration in the cell’s porous electrodes correspondingly shifts. For instance, the oxygen concentration near the cathode’s reaction sites drops noticeably during intervals of lower oxygen content in the intake air (e.g., due to high humidity or partial airflow restriction), indicating enhanced concentration polarization in the cell [37].
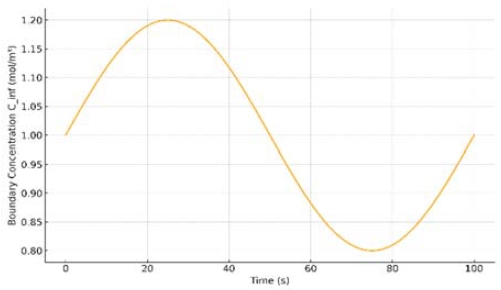
Transient boundary concentration profile (Cinf) under a sinusoidally varying marine inlet condition (baseline concentration = 1.0, amplitude ±0.2, period = 100 s), illustrating the damping effect of the porous electrode.
Conversely, when conditions improve (higher oxygen availability and optimal temperature), the concentration gradients relax, and the cell returns to near-normal operation. The porous electrode structure acts as a buffer: its tortuous gas pathways cause a slight lag in the response of the concentration field to external changes, dampening the most rapid fluctuations. Table 2 quantifies these effects by listing the range of oxygen and fuel concentrations (e.g., minimum and maximum values at critical locations or the amplitude of fluctuation) observed over the simulation period. Notably, even under aggressive environmental swings, the minimum oxygen partial pressure in the cathode remained above critical thresholds in our simulation; this suggests that short-term marine environmental fluctuations primarily lead to reversible performance effects (transient concentration losses) rather than immediate permanent damage. This finding is consistent with experimental observations that moderate drops in oxygen partial pressure cause temporary voltage loss from concentration polarization without long-term degradation, as long as the O₂ concentration does not fall below a critical limit [37]. Indeed, prior studies report that SOFCs can recover fully after brief periods of oxygen starvation if the O₂level remains above roughly 12–15% (vol.) in air whereas more severe oxygen depletion can trigger irreversible performance loss [37]. Our simulation captured this behavior: the cell endured realistic marine air fluctuations with only minor, recoverable performance variations. Overall, Figure 2 and Table 2 highlight the dynamic resilience of the SOFC’s concentration field under time-varying maritime conditions while identifying conditions that could lead to deleterious concentration polarization. These insights highlight the importance of managing air supply quality on ships–maintaining sufficient oxidant flow at all times to avoid crossing into regimes of oxygen starvation that accelerate cell deterioration.
3.3 Comparative Durability Analysis: Coastal vs. Open Sea Operational Scenarios
To evaluate long-term SOFC durability under different marine environments, the model was run for two representative scenarios: A Coastal operation (characterized by high humidity, salt spray, and variable load conditions near the port) and an Open Sea operation (open-ocean conditions with cleaner air and more stable operating parameters). Figure 3 compares the degradation of cell performance over time for these two scenarios. A clear divergence is observed: the coastal environment leads to a markedly faster performance decay than the open sea environment. In the coastal case, the cell voltage (or power output) declines more steeply as a function of operating hours, reaching the end-of-life threshold significantly sooner than in the open sea case. Table 3 summarizes key durability metrics from these simulations – for example, the time to reach a 10% voltage drop and the average degradation rate per 1000 hours in each scenario. Under coastal conditions, the model predicts a higher degradation rate (Table 3), which in our study exceeds the commonly cited durability target of about 0.2% per 1000 hours [10]. Conversely, the open sea scenario exhibits a slower degradation rate that remains closer to this target, which translates to a substantially longer service life before maintenance is required.
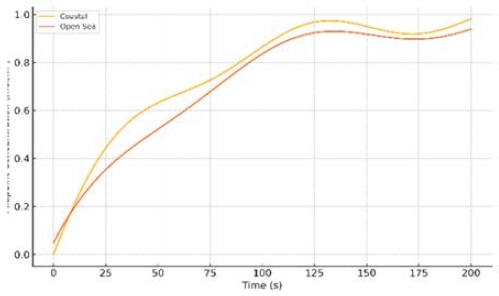
Midpoint reactant concentration over time for coastal (goldenrod) and open‐sea (coral) operating scenarios, showing accelerated depletion and recovery oscillations under coastal environmental stress

Key durability metrics for coastal vs. open sea SOFC operation. The degradation rate is defined as the percent voltage loss per 1000 h, and t₁₀% is the time to reach a 10 % voltage drop. Values are derived from long‐term dynamic simulations under representative marine boundary conditions
The accelerated degradation in the coastal scenario can be attributed to several compounding factors. First, salt and pollutants in the coastal marine air contribute to cathode contamination and material degradation. The model incorporates the effect of salt aerosol ingress, and the results align with experimental evidence that even trace levels of salt can accumulate in the cathode’s porous structure over time, leading to performance losses and eventual electrode damage [7]. In the coastal simulation, periodic exposure to salt spray (and possibly higher levels of sulfur or other airborne contaminants near the port) causes cumulative damage that manifests as a faster voltage decay. This mechanism is supported by observed failure modes in salted environments; for instance, long-term tests have shown salt deposits in the cathode can induce electrode delamination and irreversible capacity loss [7]. Second, coastal operation often involves more frequent load variations and thermal cycling (e.g., startup/shutdown cycles or load ramping when entering/exiting ports). These cyclic stresses can exacerbate degradation, as thermal and redox cycling of the cell materials are known to reduce SOFC lifespan [10]. In our coastal scenario, such stressors are reflected in the sharper performance decline. Conversely, the open sea scenario benefits from relatively steady-state operation with consistent loads and fewer environmental contaminants. The cleaner air in open waters means less impurity-induced degradation and a stable operating profile minimizes thermal cycling stress. Consequently, the open sea case maintains performance much longer, consistent with the expectation that a well-maintained SOFC in a benign environment can approach its nominal design life (often on the order of 5–7 years of continuous operation) [10][34].
It is crucial to note that while the coastal environment clearly had a more detrimental impact on the fuel cell, the model suggests that purely mechanical factors associated with the marine context (such as ship vibrations and motion) are not the primary drivers of degradation in these scenarios. This conclusion is further supported by experimental reports that ship motions (roll/pitch and vibrations) have no significant impact on SOFC lifetime when the system is properly designed for marine use [34]. Thus, the durability gap between coastal and open-sea operations in our results is predominantly due to environmental chemical stressors and operational cycling rather than the physical movement of the vessel. This finding directs attention to controlling environmental exposure as a strategy to improve SOFC longevity in maritime service.
3.4 Practical Implications for SOFC Maintenance and Operation Aboard Ships
The insights from the above results carry crucial practical implications for the deployment of SOFC systems on ships. Maintenance Scheduling: Given the accelerated degradation under coastal environmental conditions, operators should plan more frequent inspections and maintenance when ships spend extended time in coastal or port areas. For instance, the model predicts a significantly shorter time to reach critical degradation levels in coastal operation (Table 3), implying that the SOFC stack might require refurbishment or replacement earlier than the nominal schedule if exposed to harsh coastal conditions regularly. Aligning maintenance schedules with environmental exposure is thus key. In practice, this could mean scheduling stack replacements or overhauls to coincide with routine five-year dry-dock intervals for ships [34] while also performing intermediate check-ups if a vessel operates predominantly in near-shore regions. The open sea scenario, conversely, suggests that an SOFC system could potentially run for the complete intended service interval (on the order of 40,000–60,000 hours of operation) with minimal performance loss when environmental stressors are low [34]. Ship operators can utilize the predictive model to estimate cell life for specific routes; for instance, a ship that alternates between open-ocean transits and coastal operations can be assessed for its overall degradation per voyage, enabling proactive maintenance planning before failures occur.
Operational Strategies: The comparative analysis indicates that limiting the SOFC’s exposure to deleterious conditions can prolong its life. Practically, this means it may be beneficial to preferentially run the SOFC at higher loads when in clean open-sea air but operate it more conservatively (or even temporarily shut it down or run on backup systems) during periods of heavy contamination, such as in industrial port waters or during sand/dust storms in coastal regions. If shut-downs are necessary, they should be managed carefully: avoiding frequent thermal cycling will help preserve the fuel cell’s structural integrity [10]. Ideally, the SOFC system should be kept at stable operating temperature as continuously as possible to avoid stresses from heating and cooling and to prevent redox cycling of the anode (which can occur if the fuel supply is cut off at high temperature) [10]. In scenarios where a ship must port frequently, utilizing a small auxiliary power unit or battery during brief port stays (instead of repeatedly cycling the main SOFC stack on and off) could mitigate wear on the fuel cell.
Environmental Control Measures: The model results highlight the value of environmental control and system hardening for marine SOFC installations. Even trace salt contamination was identified as a serious long-term degradation driver in the coastal scenario. Therefore, integrating high-efficiency air filtration or dehumidification systems in the SOFC air intake is recommended to remove salt spray and marine aerosols before they reach the cell [7]. Similarly, using cathode air pre-heaters with desiccant filters can reduce the relative humidity and salt content of intake air, thereby minimizing the risk of salt accumulation in the cathode. In addition to air-side safeguards, ensuring fuel cleanliness is equally crucial. Marine fuels should be carefully processed to remove sulfur and other contaminants before entering the SOFC anode since sulfur poisoning can rapidly degrade anode catalysts and fuel reformers [34]. Selecting low-sulfur fuels or implementing robust desulfurization units will reduce chemical stress on the stack and extend its operational life [34].
Design Optimizations: Lastly, the findings highlight a need for marine-specific design optimizations for SOFC systems. Protective coatings on cell components (to guard against corrosion from salt), vibration-damping mounts for the stack, and enclosure systems that maintain a stable micro-climate around the SOFC can all contribute to improved durability. Indeed, previous marine trials have shown that with minor design adjustments (such as enhanced sealing and shock-resistant mounting), SOFC systems can operate reliably at sea without significant performance loss due to ship movement. Our study adds that controlling the chemical and operational environment of the SOFC is equally critical. By implementing the above strategies–environmental conditioning, careful operational scheduling, and timely maintenance–maritime operators can substantially mitigate degradation effects; this will help ensure that the SOFC system achieves its intended lifespan and performance targets in service, thereby improving the economic viability and safety of fuel cell powered ships. The predictive modeling framework developed here can serve as a decision-support tool in this process, allowing stakeholders to simulate what-if scenarios (coastal vs. open sea routes, different fuel qualities, load profiles, etc.) and optimize the SOFC usage plan for maximum durability. In summary, the combination of validated modeling and scenario analysis provides a pathway to proactively enhance SOFC longevity under real marine environmental conditions, which is essential for the successful adoption of SOFC technology in the maritime industry.
4. Conclusion
This study presented and rigorously validated a novel SOFC degradation modeling framework tailored for maritime conditions. The framework distinguishes itself through methodological originality, integrating dynamic marine environmental factors into the degradation process of the fuel cell. Unlike conventional models that assume static or land-based conditions, our approach incorporates time-varying boundary conditions (such as salt-laden humidity and temperature fluctuations) representative of at-sea operations. By calibrating the model against experimental degradation profiles, we demonstrated its ability to accurately replicate observed phenomena. This confirms both the logical integrity and physical realism of the formulation. This validated framework thus provides a reliable basis for quantifying performance decay in marine SOFC systems over time. Furthermore, the model's predictive capabilities can be extended to explore the integration and performance optimization of hybrid SOFC battery systems, significantly contributing to maritime decarbonization initiatives.
Scenario-based simulations comparing a coastal harbor environment to an open-sea environment yielded insightful findings on performance longevity. Notably, the coastal scenario showed a rapid initial accumulation of contaminants (owing to higher stagnant exposure in port), whereas the open-sea scenario, with continuous salt spray and airflow, led to a steadier but ultimately higher contaminant ingress over extended periods. These results imply that open-sea conditions may induce a more pronounced long-term degradation, even if initial deterioration is slower, whereas coastal conditions might trigger quicker early degradation that later plateaus. Such differences in the degradation trajectories underscore the importance of accounting for specific environmental contexts when predicting fuel cell lifespan. In practical terms, the model predicts that operating an SOFC system in open ocean conditions could shorten its effective service life relative to a milder coastal climate, highlighting the need for environment-tailored durability assessments in marine fuel cell deployment.
Throughout the study, the technical rigor and predictive strength of the model have been evident. The model is built on fundamental transport and electrochemical degradation principles, ensuring that its behavior remains consistent with known physical laws under all tested scenarios. Extensive calibration and sensitivity analysis have reinforced the model’s robustness, and the close agreement between simulated outcomes and experimental data attests to its accuracy. Furthermore, the framework’s capacity to predict varying degradation patterns across different scenarios underscores its exceptional predictive power for practical applications. The logical coherence of the model–from its assumptions to its computational implementation–instills confidence that the predictions are not artifacts of numerical choices but true reflections of degradation dynamics.
This work makes a significant scholarly contribution to the field of marine SOFC modeling by integrating environmental engineering with fuel cell degradation science. It stands out as one of the pioneering studies that explicitly simulates and compares SOFC durability under realistic maritime scenarios, thereby filling a critical gap in the literature. The findings offer original insights into how marine environmental stressors–particularly salt spray and coastal vs. open-sea conditions–impact long-term fuel cell performance. Overall, the study advances academic understanding of SOFC longevity in marine applications and establishes a robust, predictive modeling approach that future researchers and engineers can build upon in the quest for durable, ocean-worthy fuel cell systems.
5. Future Work
The present calibration is anchored in two data streams: (i) laboratory button-cell tests reported in public degradation studies and (ii) 5–10 kW auxiliary-power-unit (APU) stacks installed on short-route coastal ferries. Although these sources capture key maritime stressors, they do not yet encompass propulsion-class SOFC modules (> 500 kW) or very large vessels such as LNG carriers. Consequently, extrapolation of the current parameter set to all ship types should be approached with caution. Future work will focus on three priorities: 1) validating the framework against full-scale sea trials covering cargo ships, cruise vessels, and offshore platforms; 2) extending the load library to include multi-route duty cycles that span polar, temperate, and tropical climates; and 3) embedding six-degree-of-freedom ship-motion data (heave, pitch, roll, surge, sway, yaw) into the thermo-mechanical solver to quantify vibration- and sloshing-induced degradation. These enhancements will support vessel-specific durability assessments and accelerate the deployment of SOFC technology across the broader maritime sector.
Acknowledgments
This research was supported by a grant from the National R&D Project “Research Hub Establishment and International Joint Research on Advanced Maritime Mobility (AMM) between Korea and Europe” funded by the Ministry of Oceans and Fisheries (RS-2024-0041020).
Author Contributions
Conceptualization, J. K. Lee; Methodology, E. J. Park; Software, E. J. Park; Formal Analysis, E. J. Park and J. K. Lee; Investigation, E. J. Park; Resources, E. J. Park; Data Curation, E. J. Park; Writing-Original Draft Preparation, E. J. Park; Writing-Review & Editing, J. K. Lee; Visualization, E. J. Park and J. K. Lee; Supervision, J. K. Lee; Project Administration, J. K. Lee; Funding Acquisition, J. K. Lee
References
- International Maritime Organization (IMO), Fourth IMO Greenhouse Gas Study 2020, IMO Publications, London, UK, 2021.
- International Maritime Organization (IMO), IMO Strategy on Reduction of GHG Emissions from Ships, Resolution MEPC.377(80), London, 2023.
-
B. N. van Veldhuizen, L. van Biert, P. V. Aravind, and K. Visser, “Solid oxide fuel cells for marine applications,” International Journal of Energy Research, vol. 2023, Art. ID 5163448, 2023.
[https://doi.org/10.1155/2023/5163448]

-
B. V. Sasank, N. Rajalakshmi, and K. S. Dhathathreyan, “Performance analysis of polymer electrolyte membrane fuel cell stack operated under marine environmental conditions,” Journal of Marine Science and Technology, vol. 21, no. 4, pp. 471–478, 2016.
[https://doi.org/10.1007/s00773-016-0369-y]

-
S. Mekhilef, R. Saidur, and A. Safari, “Comparative study of different fuel cell technologies,” Renewable and Sustainable Energy Reviews, vol. 16, no. 1, pp. 981–989, 2012.
[https://doi.org/10.1016/j.rser.2011.09.020]

-
V. Venkataraman, M. Pérez‑Fortes, L. Wang, Y. S. Hajimolana, C. Boigues‑Muñoz, C. Agostini, S. J. McPhail, F. Maréchal, J. Van Herle, and P. V. Aravind, “Reversible solid oxide systems for energy and chemical applications—review & perspectives,” Journal of Energy Storage, vol. 24, Art. ID 100782, 2019.
[https://doi.org/10.1016/j.est.2019.100782]

-
N. H. Menzler, D. Sebold, Y. J. Sohn, and S. Zischke, “Post-test characterization of a solid oxide fuel cell after more than 10 years of stack testing,” Journal of Power Sources, vol. 478, Art. ID 228770, 2020.
[https://doi.org/10.1016/j.jpowsour.2020.228770]

-
P. Aguiar, C. S. Adjiman, and N. P. Brandon, “Anode-supported intermediate temperature direct internal reforming solid oxide fuel cell. i: model-based steady-state performance,” Journal of Power Sources, vol. 138, no. 1–2, pp. 120–136, 2004.
[https://doi.org/10.1016/j.jpowsour.2004.06.040]

-
M. B. Mogensen, A. Hauch, X. Sun, M. Chen, Y. Tao, S. D. Ebbesen, K. V. Hansen, and P. V. Hendriksen, “Relation between Ni particle shape change and Ni migration in Ni–YSZ electrodes—a hypothesis,” Fuel Cells, vol. 17, no. 4, pp. 434–441, 2017.
[https://doi.org/10.1002/fuce.201600222]

-
H. S. Lee, H. M. Lee, J. -Y. Park, and H. -T. Lim, “Degradation behavior of Ni–YSZ anode-supported Solid Oxide Fuel Cell (SOFC) as a function of H2S concentration,” International Journal of Hydrogen Energy, vol. 43, no. 49, pp. 22511–22518, 2018.
[https://doi.org/10.1016/j.ijhydene.2018.09.189]

-
M. Hubert, J. Laurencin, P. Cloetens, J. Mougin, R. Porcaro, D. Rouchon, and B. Verdier, “Solid oxide cell degradation operated in fuel cell and electrolysis modes: a comparative study on Ni agglomeration and LSCF destabilization,” ECS Transactions, vol. 78, no. 1, pp. 3167–3177, 2017.
[https://doi.org/10.1149/07801.3167ecst]

-
H. Kurokawa, C. Jacobson, L. DeJonghe, and S. Visco, “Chromium vaporization of bare and of coated iron–chromium alloys at 1073 K,” Solid State Ionics, vol. 178, no. 3–4, pp. 287–296, 2007.
[https://doi.org/10.1016/j.ssi.2006.12.010]

-
M. Kornely, A. Neumann, N. H. Menzler, A. Leonide, A. Weber, and E. Ivers‑Tiffée, “Degradation of Anode Supported Cell (ASC) performance by Cr-poisoning,” Journal of Power Sources, vol. 196, no. 17, pp. 7203–7208, 2011.
[https://doi.org/10.1016/j.jpowsour.2010.10.033]

-
S. Fontana, R. Amendola, S. Chevalier, P. Piccardo, G. Caboche, M. Viviani, R. Molins, M. Sennour, “Metallic interconnects for SOFC: characterisation of corrosion resistance and conductivity evaluation at operating temperature of differently coated alloys,” Journal of Power Sources, vol. 171, no. 2, pp. 652–662, 2007.
[https://doi.org/10.1016/j.jpowsour.2007.06.255]

-
J. W. Stevenson, Z. G. Yang, G. G. Xia, Z. Nie, J. D. Templeton, “Long-term oxidation behavior of spinel-coated ferritic stainless steel for solid oxide fuel cell interconnect applications,” Journal of Power Sources, vol. 231, pp. 256–263, 2013.
[https://doi.org/10.1016/j.jpowsour.2013.01.033]

- S, -I. Lee, J. Hong, H. Kim, J. W. Son, J. -H. Lee, B. -K. Kim, H. -W. Lee, and K. J. Yoon, “Highly dense Mn–Co spinel coating for protection of metallic interconnect of solid oxide fuel cells,” Journal of The Electrochemical Society, vol. 161, no. 14, pp. F1389–F1394, 2014.
-
B. Talic, S. Molin, K. Wiik, P. V. Hendriksen, and H. L. Lein, “Comparison of iron and copper doped manganese cobalt spinel oxides as protective coatings for solid oxide fuel cell interconnects,” Journal of Power Sources, vol. 372, pp. 145–156, 2017.
[https://doi.org/10.1016/j.jpowsour.2017.10.060]

-
M. Guida, F. Postiglione, and G. Pulcini, “A random-effects model for long-term degradation analysis of solid oxide fuel cells,” Reliability Engineering & System Safety, vol. 140, pp. 88–98, 2015.
[https://doi.org/10.1016/j.ress.2015.03.036]

-
E. Audasso, F. R. Bianchi, and B. Bosio, “2D Simulation for CH4 internal reforming-SOFCs: An approach to study performance degradation and optimization,” Energies, vol. 13, no. 16, Art. ID 4116, 2020.
[https://doi.org/10.3390/en13164116]

-
R. Spotorno, F. R. Bianchi, D. Paravidino, B. Bosio, and P. Piccardo, “Test and modelling of solid oxide fuel cell durability: A focus on interconnect role on global degradation,” Energies, vol. 15, no. 8, Art. ID 2762, 2022.
[https://doi.org/10.3390/en15082762]

- K. Kendall and S. C. Singhal, High-Temperature Solid Oxide Fuel Cells: Fundamentals, Design and Applications, Elsevier, Amsterdam, 2003.
- N. Q. Minh and T. Takahashi, Science and Technology of Ceramic Fuel Cells, Elsevier, Amsterdam, 1995.
-
V. M. Janardhanan and O. Deutschmann, “Modeling diffusion limitation in solid-oxide fuel cells,” Electrochimica Acta, vol. 56, no. 27, pp. 9775–9782, 2011.
[https://doi.org/10.1016/j.electacta.2011.08.038]

- S. B. Adler, “Limitations of charge-transfer models for mixed-conducting oxygen electrodes,” Solid State Ionics, vol. 135, no. 1–4, pp. 603–612, 2000.
-
J. Kim, E. Ali, M. Kim, H. Lim, and H. Park, “Effect of water vapor generation on cell polarization in active area of anode-supported solid oxide fuel cells,” Heat and Mass Transfer, vol. 56, no. 2, pp. 617–625, 2020.
[https://doi.org/10.1007/s00231-019-02732-9]

- D. Mogensen, J. -D. Grunwaldt, P. V. Hendriksen, K. Dam-Johansen, J. U. Nielsen, “Internal steam reforming in solid oxide fuel cells: status and opportunities of kinetic studies and their impact on modelling,” Journal of Power Sources, vol. 196, no. 1, pp. 25–38, 2011.
-
O. B. Rizvandi, X. -Y. Miao, and H. L. Frandsen, “Multiscale modeling of degradation of full solid oxide fuel cell stacks,” International Journal of Hydrogen Energy, vol. 46, no. 54, pp. 27709–27730, 2021.
[https://doi.org/10.1016/j.ijhydene.2021.05.204]

-
J. W. Fergus, “Electrolytes for solid oxide fuel cells,” Journal of Power Sources, vol. 162, no. 1, pp. 30–40, 2006.
[https://doi.org/10.1016/j.jpowsour.2006.06.062]

-
H. Kurokawa, C. Jacobson, L. DeJonghe, and S. Visco, “Chromium Vaporization of Bare and of Coated Iron–Chromium Alloys at 1073 K,” Solid State Ionics, vol. 178, no. 3–4, pp. 287–296, 2007.
[https://doi.org/10.1016/j.ssi.2006.12.010]

- Z. Lyu, H. Li, Y. Wang, and M. Han, “Performance degradation of solid oxide fuel cells analyzed by evolution of electrode processes under polarization,” Journal of Power Sources, vol. 485, Art. ID 229237, 2021.
-
S. P. Jiang, “A review of wet impregnation – an alternative method for the fabrication of high-performance and nano-structured electrodes of solid oxide fuel cells,” Materials Science and Engineering A, vol. 418, no. 1–2, pp. 199–210, 2006.
[https://doi.org/10.1016/j.msea.2005.11.052]

- D. Lee, J. -K. Lin, C. -H. Tsai, S. -H. Wu, Y. -N. Cheng, R. -Y. Lee, “Analysis of long-term and thermal cycling tests for a commercial solid oxide fuel cell,” Journal of Electrochemical Energy Conversion and Storage, vol. 14, no. 4, Art. ID 041002, 2017.
-
F. Alenazey, Y. Alyousef, B. AlOtaibi, G. Almutairi, M. Minakshi, C. K. Cheng, and D. -V. N. Vo, “Degradation behaviors of solid oxide fuel cell stacks in steady-state and cycling conditions,” Energy & Fuels, vol. 34, no. 11, pp. 14864–14873, 2020.
[https://doi.org/10.1021/acs.energyfuels.0c02920]

-
N. Thambiraj, I. Wærnhus, C. Suciu, A. Vik, and A. C. Hoffmann, “Single-cell tests to explore the reliability of SOFC installations operating offshore,” Energies, vol. 13, no. 7, Art. ID 1624, 2020.
[https://doi.org/10.3390/en13071624]

-
S. Yang, Z. Chen, H. Zhang, J. Li, X. Zhao, W. Hao, J. Xie, F. Wang, “Failure analysis of Ni–8YSZ electrode under re-oxidation based on the real microstructure,” Materials, vol. 17, no. 18, Art. ID 4599, 2024.
[https://doi.org/10.3390/ma17184599]

- C. Yang, R. Guo, X. Jing, P. Li, J. Yuan, and Y. Wu, “Degradation mechanism and modeling study on reversible solid oxide cell in dual-mode—A review,” International Journal of Hydrogen Energy, vol. 47, no. 89, pp. 37895–37928, 2022.
-
A. Faes, A. Nakajo, A. Hessler-Wyser, D. Dubois, A. Brisse, S. Modena, and J. V. Herle, “RedOx study of anode-supported solid oxide fuel cells,” Journal of Power Sources, vol. 193, no. 1, pp. 55-64, 2009.
[https://doi.org/10.1016/j.jpowsour.2008.12.118]